Principle
A sandwich based ligand binding assay was developed by Triangle Laboratory for the quantitation of RF001 in human serum (Figure1). In brief, validation samples are diluted and transferred into MSD plate, which pre-adsorbs Capture antibody (Clone#D5B). After applying detection antibody (Ruthenylated monoclonal antibody, Clone# 9F3), the plate is read using MSD Sector Imager S600.
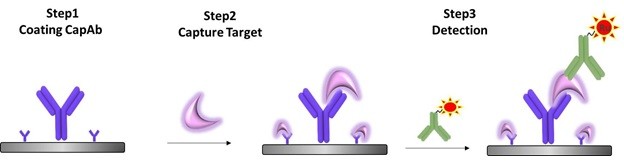
Figure1: Scheme of sandwich based ligand binding assay
Challenge
Client required the sensitivity should be as low as 10ng/mL and provided our team with one polyclonal and four kinds of monoclonal antibodies, different binding affinity of antibody with target will determine the final sensitivity of assay method. So orthogonal experiment design will be applied in the method development. The other key point is how to choose the suitable blocking buffer and binding buffer to obtain the stable electrochemiluminescence signal in human serum samples. The most important thing is how to set PK linearity range in order to avoid the curve plateau, which would build analytical method with good accuracy and high reproducibility (Figure2).
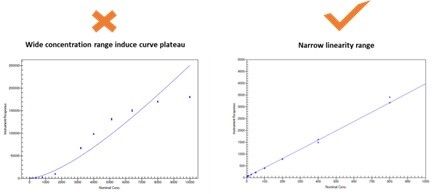
Figure2: Optimization of PK linearity range
Our Solution
The scientists in our Bio-macromolecular team discussed assay idea and progress with client. First, orthogonal experiment design was employed in the selection of capture antibody and detection antibody, four pairs of combination antibodies were tested with different binding buffers.
Then different Minimum Required Diluent (MRD) was test with pooled human serum, in order to get the low background and the optimal signal to noise(S/N). Finally, LLOQ of 10ng/mL was achieved successfully and the stability of sample is good on bench-top and freeze thaw for 6 cycles, also the intra-batch and inter-batch accuracy and precision were acceptable for all QC samples, the robustness of the current developed method is good. Also the specificity of this method will be tested with interference drug based on clinical requirement.
